Rapid Whole Genome Sequencing: The High-Tech Solution to Lowering Infant Mortality (And Reducing Medical Costs, Too)
This piece reflects the opinions of the author.
Genomic testing can drastically reduce the waiting time in the diagnosis of genetic disorders, not only saving lives, but money. These services, including rapid whole genome sequencing (rWGS), should be a standard of care for infants presenting with symptoms or screening results that suggest the possibility of a deadly or debilitating condition.
The infant mortality rate (IMR) in the United States is 5.7 per 1,000 births, which places our nation near the bottom (33rd) when ranked against 37 other OECD countries. In the United States, the top reasons for infant death and hospitalization are congenital anomalies and genetic disorders, affecting approximately 6% of all live births. The initial diagnosis of these genetic disorders in infants can be challenging, often due to the complex array of symptoms that can be associated with these conditions.
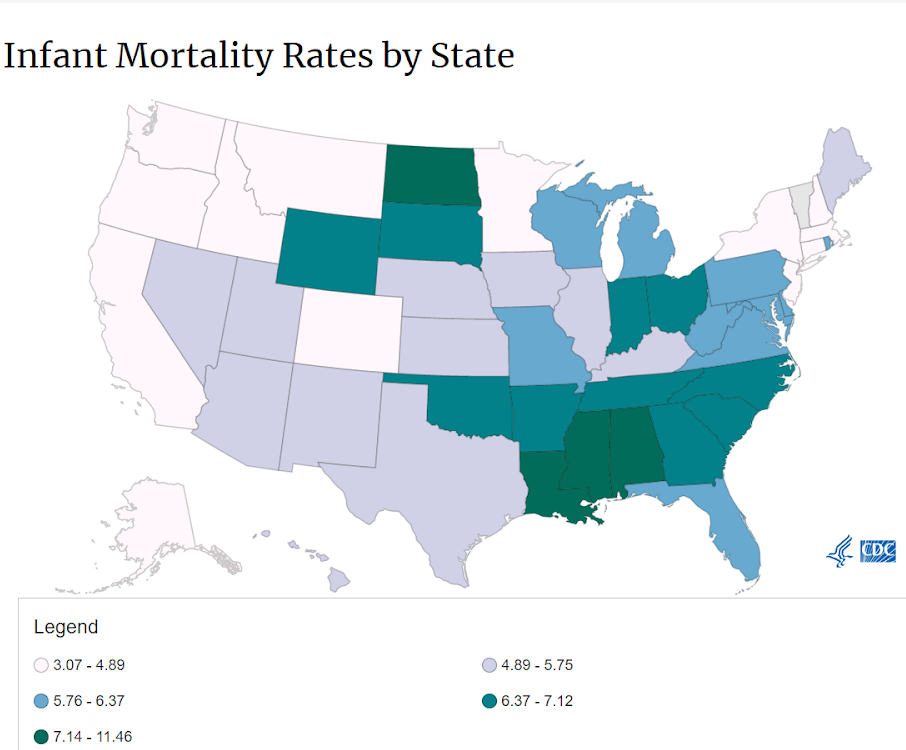 (CDC, 2018)
(CDC, 2018)
Whole genome sequencing has contributed to improving infant health and provides promise for developing new therapies, while also helping doctors make sense of ambiguous or atypical symptoms. This groundbreaking technology establishes the entire DNA sequence with one analysis. While standard sequencing usually takes weeks, rapid whole genome sequencing (rWGS) provides the same crucial information in a fraction of the time.
Both sequencing methods have been used in neonatal and pediatric intensive care units (ICU) to diagnose genetic abnormalities/disorders, thus providing targeted patient management, guiding the most optimal medical and surgical treatment options, and improving overall infant health. The agility of rWGS has the potential to make revolutionary strides in medical care for infants, and it should be implemented as routine care policy for critically and acutely ill infants admitted into the ICU.
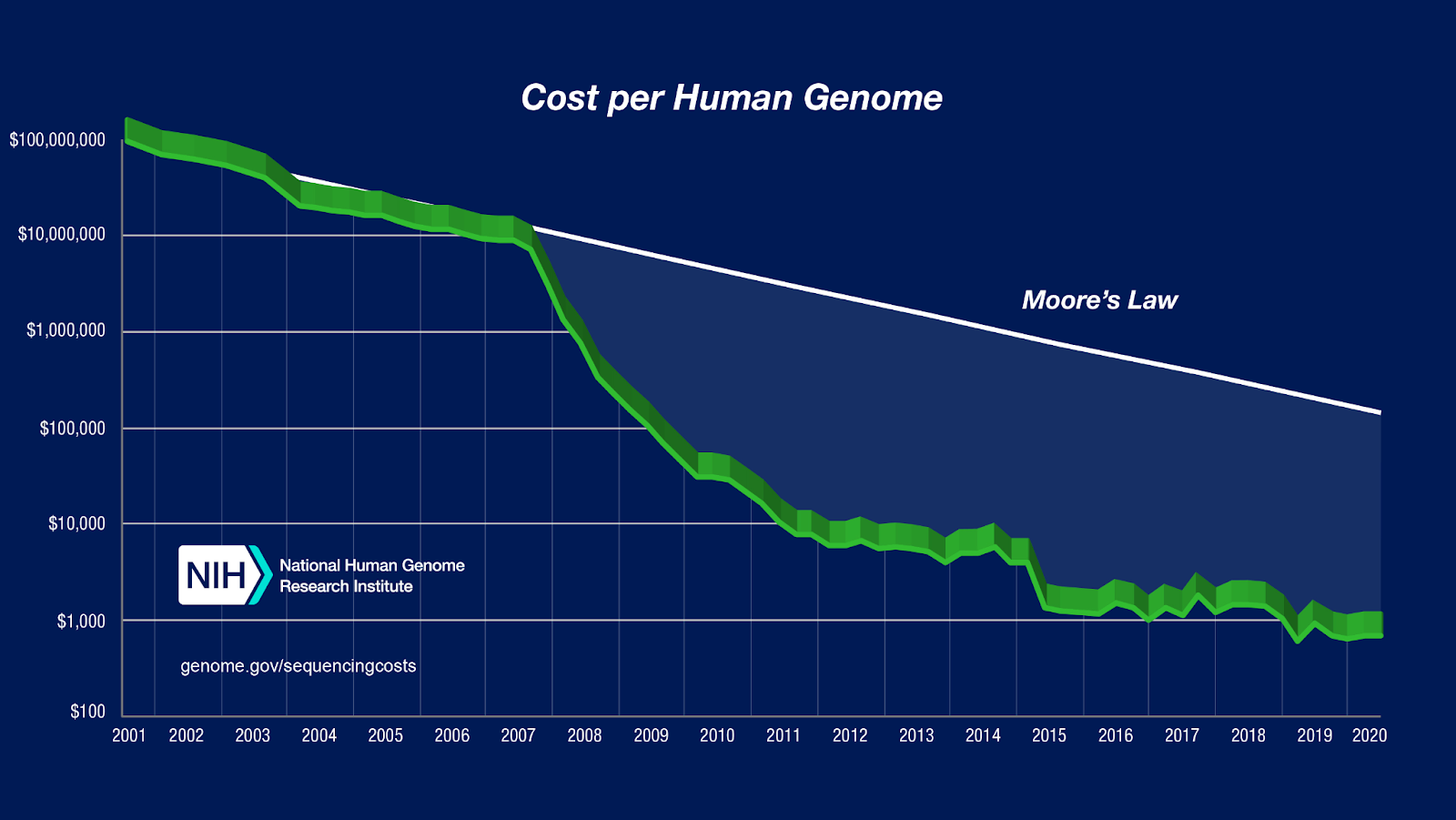
One expert shared that a single rWGS explores 6,000 genetic diseases in 13.5 hours and costs $700 (scientists predict that the cost of rWGs will be as low as $10 per patient by 2030).
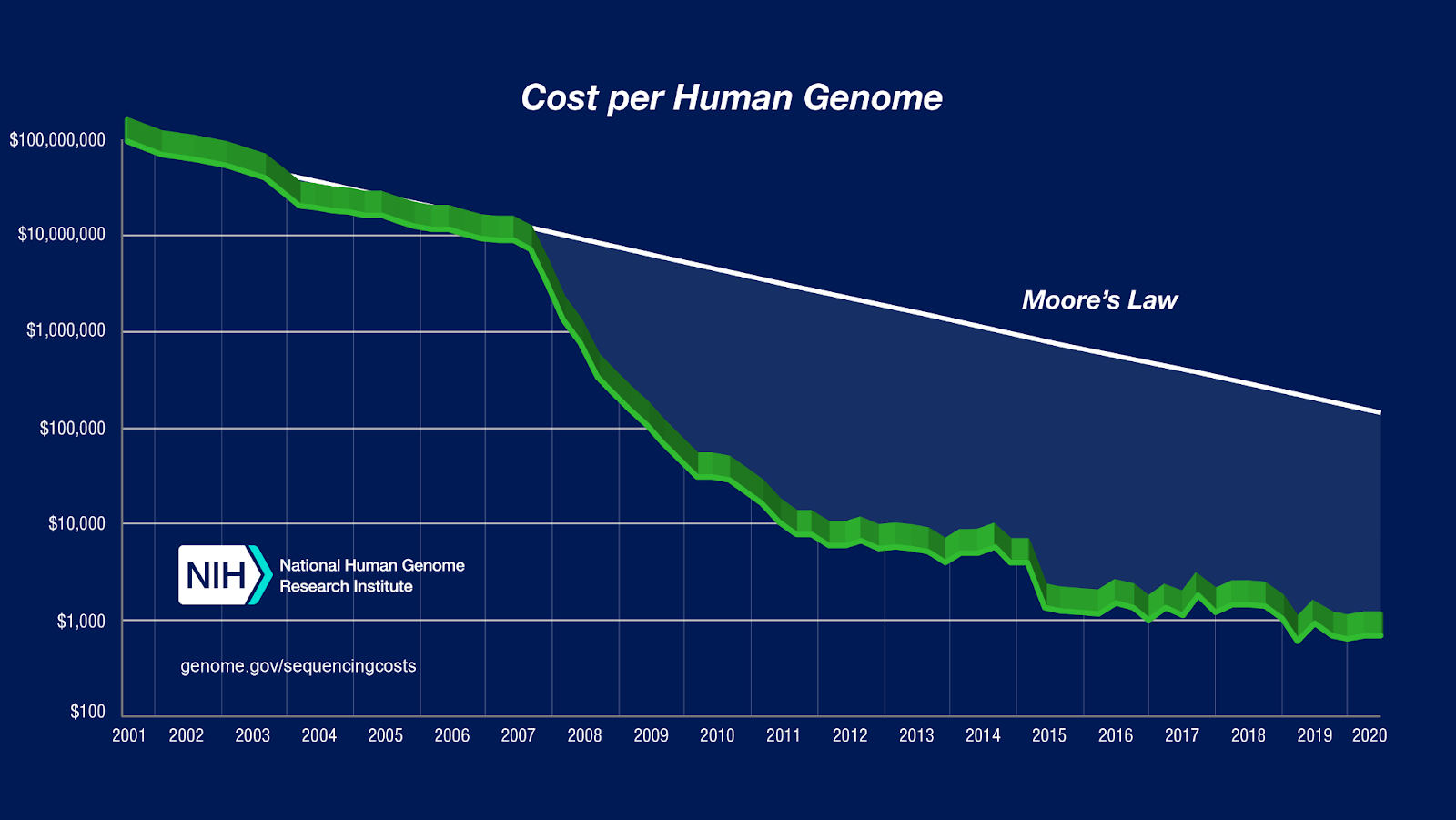
(CDC, 2020)
There are several clinical examples that support the use of rWGS in critically and acutely ill infants. Between July 2016 and March 2017, 42 infants in the United States received rWGS, leading to diagnosis of genetic disorders. The rate of clinical utility (positive change in medical or surgical management) was 31% (13 of 42 infants), and avoided morbidity was 26% (11 of 42 infants). Six of the 11 infants that avoided morbidity received changes in management that reduced inpatient cost by $800,000–$2,000,000. The estimated cost of applying rWGS for all 42 infants was $29,000–$31,000—well under the reduced inpatient cost savings.
A more recent study evaluated 25 critically ill infants in UCSF Benioff Children’s Hospital San Francisco’s Newborn Intensive Care Unit (NICU) who underwent rWGS. Within 48 hours, 16 cases revealed genetic diagnoses and 14 infants had a change in care management. A study from the Centers for Disease Control and Prevention found that the average diagnosis time for rWGS is approximately four days, and the average diagnosis time for standard genome sequencing can take up to 42 days. While rWGS is more costly compared to standard genome sequencing, avoiding 38 days in the hospital can save an estimated $181,141 (including the cost of rWGS) and prevent painful/risky procedures.
Health and government leaders in California have taken initiative to pave the way for implementing routine rWGS policy in pediatric ICUs. In 2018, five hospitals in the state provided rWGS to all infants admitted into the ICU unit. Over the span of three days, 40% of infants received a diagnosis (most of which were rare disorders) and one-third of infants received more personalized medical care. With a goal of strengthening infant health, Assemblymember Brian Maienschein, representing the 77th California Assembly District, authored and introduced the AB-114 Medi-Cal benefits: Rapid Whole Genome Sequencing (2020–2021), which would include rWGS for any Medi-Cal beneficiary who is one year of age or younger and is receiving inpatient hospital services in the ICU. In March 2020, Blue Shield of California became the first health plan in the United States to cover rWGS (and ultra-rWGS) to help critically ill infants in the ICU with unexplained medical conditions receive personalized care.
The application of rGWS should become a standard for critically ill infants in PICU and NICU settings. Current findings from rWGS implementation have shown a significant improvement in infant health that saves both time and money. Through greater use of rGWS, infant deaths from congenital malformations, deformations, chromosomal abnormalities, and other disorders can be significantly lowered. Data from rWGS application in infants will provide opportunities for research, allow scientists to analyze genetic diseases at a larger scale, create models for infant care, and uncover insights that will undoubtedly lead to improved pediatric health and a reduction in the nation’s infant mortality rate.
Ashni Dhruva is a former science policy intern at Research!America.




