Tax Money Spent Wisely
 Yesterday was Tax Day, a date everyone knows as the deadline for filing their tax returns. This year, it’s estimated that Americans will spend $146 billion in time and out-of-pocket expenses (subscription required) to file their taxes. That’s almost double the amount that was appropriated for crucial health and research agencies for FY26.
Yesterday was Tax Day, a date everyone knows as the deadline for filing their tax returns. This year, it’s estimated that Americans will spend $146 billion in time and out-of-pocket expenses (subscription required) to file their taxes. That’s almost double the amount that was appropriated for crucial health and research agencies for FY26.
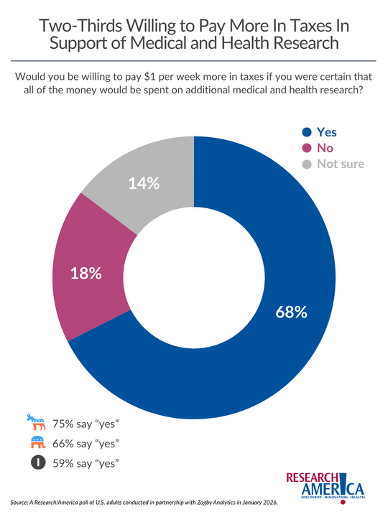
And speaking of taxes, over the past few years, our commissioned surveys have asked respondents whether they would be willing to pay $1 more per week in taxes if they were certain that money would be spent on additional medical and health research. More than two-thirds (68%) of Americans said yes.
On the Hill: Congress is moving ahead with the FY27 appropriations process. The House Appropriations Committee has released its markup schedule, with the Labor-HHS Subcommittee markup currently set for June 5, followed by full committee consideration on June 9. As a reminder, a markup is when members of Congress debate, amend, and vote on a draft funding bill before it advances to the next stage. To learn more about House markups, read this CRS report here.
(Please note, the timeline may shift as the process moves forward; we’ll keep you posted on any changes and key opportunities for engagement.)
CDC Director Nominee Announced: President Donald Trump has announced the nomination of Dr. Erica Schwartz as director of the Centers for Disease Control and Prevention. Dr. Schwartz, who previously served in the U.S. military and as Deputy Surgeon General during the first Trump Administration, holds degrees in medicine, law, and public health.
Hearings and More Hearings:
- This week, both House and Senate (view the recordings) Budget Committees held hearings on the President’s FY27 budget proposal, with Office of Management and Budget (OMB) Director Russ Vought testifying. Research!America submitted a statement for the record underscoring the need for sustained investment in medical and public health research; read our statement here.
- HHS Secretary Kennedy was on Capitol Hill this week discussing the budget with the House Appropriations Labor-HHS Subcommittee and the House Ways and Means Committee (view the recordings). The hearings featured pointed exchanges with lawmakers over proposed funding cuts, vaccine policy, and broader public health priorities, with the Secretary emphasizing cost-cutting and a shift toward prevention-focused care. Secretary Kennedy will continue testifying next week. Upcoming hearings, which you can watch here:
Indirect Cost Reimbursement: Last week’s letter covered the administration’s decision to end its legal efforts to establish a 15% cap on indirect cost reimbursement. A Washington Post opinion piece(subscription required) published earlier this week put a fine point on the damage such a cap would do and referenced alternate reimbursement approaches such as the FAIR model. Note that the administration included the 15% cap in its recent FY27 budget proposal, which reportedly they plan to accomplish via a combination regulatory/statutory strategy. More to come on this in future letters!
SBIR/STTR Reauthorized! On April 13, President Trump signed into law the Small Business Innovation and Economic Security Act (S. 3971), reauthorizing the Small Business Innovation Research (SBIR) and Small Business Technology Transfer (STTR) programs through Sept. 30, 2031. The programs, which had lapsed on Sept. 30, 2025, are often referred to as “America’s Seed Fund.” These programs have invested more than $81 billion in more than 34,000 small businesses since 1982, helping launch new companies and leveraging billions in follow-on private investment to bring new technologies to market across sectors.
NSF to Study Compensation for Early-career Researchers: The National Science Foundation (NSF) has agreed to establish a study on graduate student compensation, as recommended by the Government Accountability Office’s report of April 8. Centralized data on graduate researcher compensation does not exist, the GAO report found. The report also analyzed cost of living, grant funding stability, and other factors that influence these researchers’ decisions to pursue and remain in federally funded research programs.
Mark Your Calendars:
Annual Meeting: Next week, we will host Research!America’s Annual Meeting of Members. On April 23, from 1-2:15 p.m. ET members will vote to approve new and renewed board members, and National Cancer Institute Director Dr. Anthony Letai will deliver remarks. If your organization is a Research!America alliance member, check your inbox for registration information or reach out to Jasmine George.
Other Special Events:
- Webinar on FDA Rare Disease Innovation Hub: We are pleased to host Amy Comstock Rick, Associate Director for Rare Disease Strategy at the Center for Drug Evaluation and Research (CDER) at FDA and Director of Strategic Coalitions for the Rare Disease Innovation Hub, on Thursday, April 30, at noon ET on Zoom. Amy, who is an emeritus board member of Research!America, will discuss FDA’s work to advance treatments for people living with rare diseases. This webinar is open to the public. Register here.
- Special Alliance Member-only Meeting: Join us for a special member-only Zoom discussion on Thursday, April 23, from 11:30 a.m. to 12:15 p.m. ET featuring Meghan Mott and Stephen Steigleder, Professional Staff Members for the Minority on the Senate and House Labor-HHS Subcommittees. We’ve asked Meghan and Stephen to provide an update on the FY27 appropriations process, with a particular focus on NIH. Registration is limited to individuals from Research!America member organizations. Please watch your email for an invitation or email Jacqueline Lagoy. Interested in joining the alliance? Contact Donald Cook.
Support Our Advocacy Work: Your support is critical for our advocacy efforts. Research!America is working every day to ensure our nation’s discovery, development, and innovation ecosystem not only survives, but thrives. Working together, we can make medical and public health as well as overall science and technology progress a higher national priority. Donate now.




