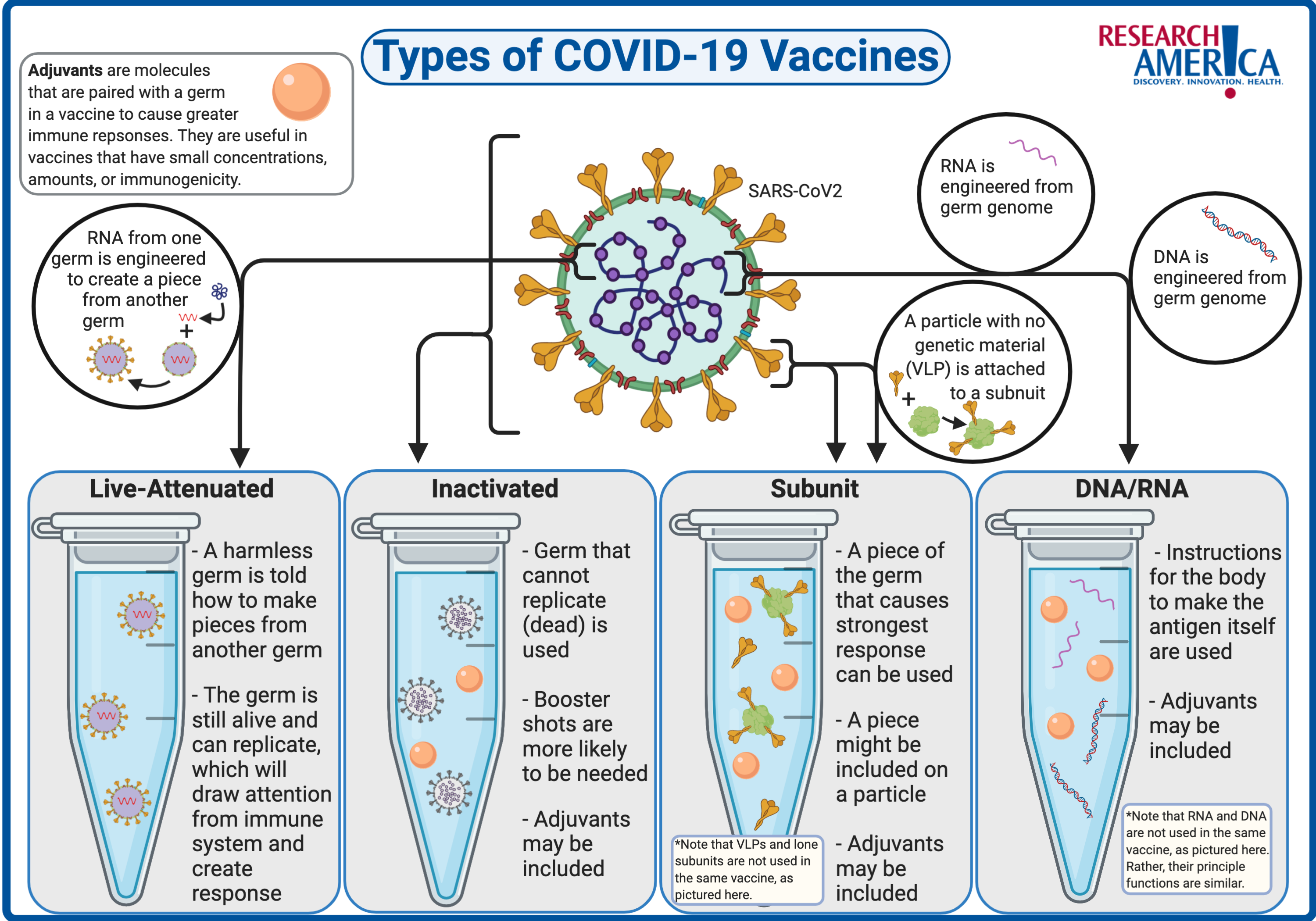
Types of Vaccines

As scientists work towards a COVID-19 vaccine, they are faced with the decision of which type of vaccine to choose. Here, we review the different types of vaccines, how they work, some examples, and current COVID-19 trials in each category. For additional information on the science behind vaccine approaches for covid-19, take a look at Research!America’s COVID-19: By the Science infographic.
Live-attenuated vaccines
Live-attenuated vaccines use a weakened form of the germ (bacteria or virus) that causes the disease. Scientists weaken the germ by altering its genetic code to prevent the germ from rapidly replicating in human cells. This weakened form can no longer cause the disease, but the immune system is able to see what the virus looks like and create a long-lasting immune response.8 For COVID-19 vaccines, most scientists are not focusing on live-attenuated vaccines that contain the SARS-CoV2 virus. Instead, they are testing previously generated live-attenuated viruses (such as the common cold and the measles virus) but are manipulating the genetic code of the virus to display the key SARS-CoV2 spike protein.9 This type of vaccine is a subtype of live-attenuated vaccines known as a viral-vector vaccine.
Examples: MMR, Rotavirus, Chickenpox, Yellow fever
COVID-19 examples:
Measles Vector Vaccine in preclinical trials
(University of Pittsburgh’s Center for Vaccine Research)
AZD1222 (Adenovirus a.k.a. common cold virus) in phase 2/3
(The University of Oxford / AstraZeneca)
Ad5-nCoV (Adenovirus a.k.a. common cold virus) in phase 2
(CanSino)
Inactivated vaccines
Inactivated vaccines use a dead form of the germ that causes the disease. Scientists kill the germ using heat and/or chemicals. Just like the live-attenuated vaccine, the inactivated germ is able to show the immune system what the germ looks like without making the patient sick. Normally, an inactivated vaccine is not as strong as the live-attenuated vaccines and may require booster shots over time.8 Current clinical trials of inactivated COVID-19 vaccines are testing inactivated SARS-CoV2 virus.
Examples: Hepatitis A, Flu, Polio, Rabies
COVID-19 Example:
Inactivated vaccine in phase 3
(Wuhan Institute of Biological Products et al.)
CoronaVac in phase 3
(Sinovac)
Subunit vaccines
When it comes down to it, the immune system really only needs to be shown a key piece of the germ, not the whole thing, in order to develop a strong immune response. Subunit vaccines use a piece — protein or sugar — that is part of the germ that causes the disease. Usually, this piece is on the exterior of the germ; however, identifying and producing the correct piece is not trivial. If the correct piece can be identified, these vaccines are often highly effective.8 For the COVID-19 subunit vaccines, researchers are focusing on the spike, “S” protein which sticks out on the surface of the SARS-CoV2 virus. Additionally, some scientists are investigating specific subunit vaccines that use virus-like particles (VLP) to display the subunit to the immune system.9 These particles have a membrane, similar to the actual SARS-CoV2 virus, but no genetic code within the particles — meaning they are not infectious. The particles hold many subunits together helping the immune system identify the subunit.
Examples: Hepatitis B, HPV, Shingles
COVID-19 Example:
Ad26.COV2-S in phase 1/2
(Johnson & Johnson)
PittCoVacc in preclinical trials
(University of Pittsburgh)
Virus-Like Particle Vaccine in phase 1
(Medicago)
DNA/RNA Vaccines
In a cell, DNA is transcribed into RNA, which is then translated into protein. A DNA or RNA vaccine is essentially a subunit vaccine, but instead of introducing a subunit (protein) into the body, this vaccine provides the specific genetic code (DNA or RNA) for the cells which produce the subunit itself.10 The benefit is that RNA or DNA is more easily and inexpensively produced in a laboratory compared to producing a subunit. RNA vaccines have the added benefit of no risk that the RNA will integrate into the patient’s own genetic code — as could happen with a DNA vaccine. There are currently no DNA or RNA vaccines used in the United States, predominantly because the technology is new.11 These vaccines present a relatively simple and exciting way to develop a COVID-19 vaccine.
DNA COVID-19 Example:
INO-4800 in phase 1
(Inovio Pharmaceuticals)
GX-19 in phase 1/2
(Genexime)
RNA COVID-19 Example:
mRNA-1273 in phase 2, set to enter phase 3 in July 2020
(Moderna)
BNT162 in phase 1/2
(Pfizer and BioNTech)
For a list of current COVID-19 vaccine trials click here.